|
Specific Lesson Objectives:
a) state the symbols of the elements and formulae of the compounds mentioned in the syllabus b) deduce the formulae of simple compounds from the relative numbers of atoms present and vice versa c) deduce the formulae of ionic compounds from the charges on the ions present and vice versa d) interpret chemical equations with state symbols |
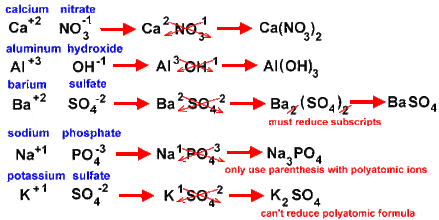
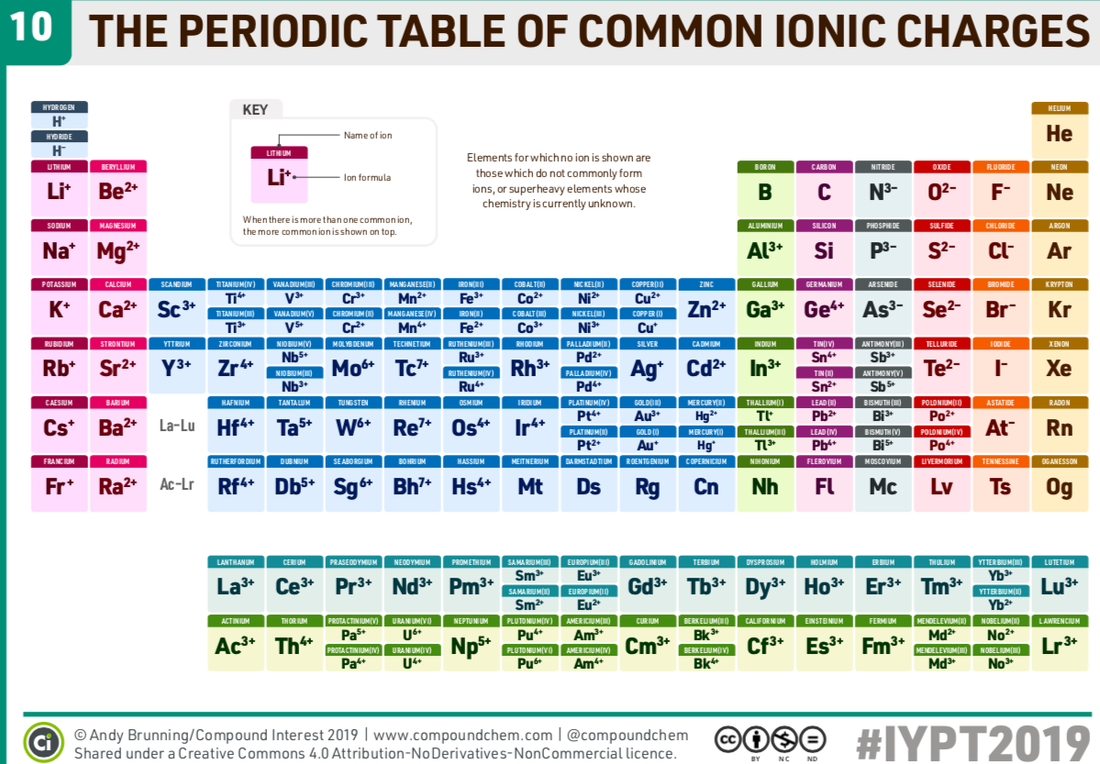
The Criss-Cross Method to write Chemical Formulas.
|
Self-directed Learning
Writing Chemical Formula
|
Step 1:
Write the correct chemical formula for the reactants and products. |
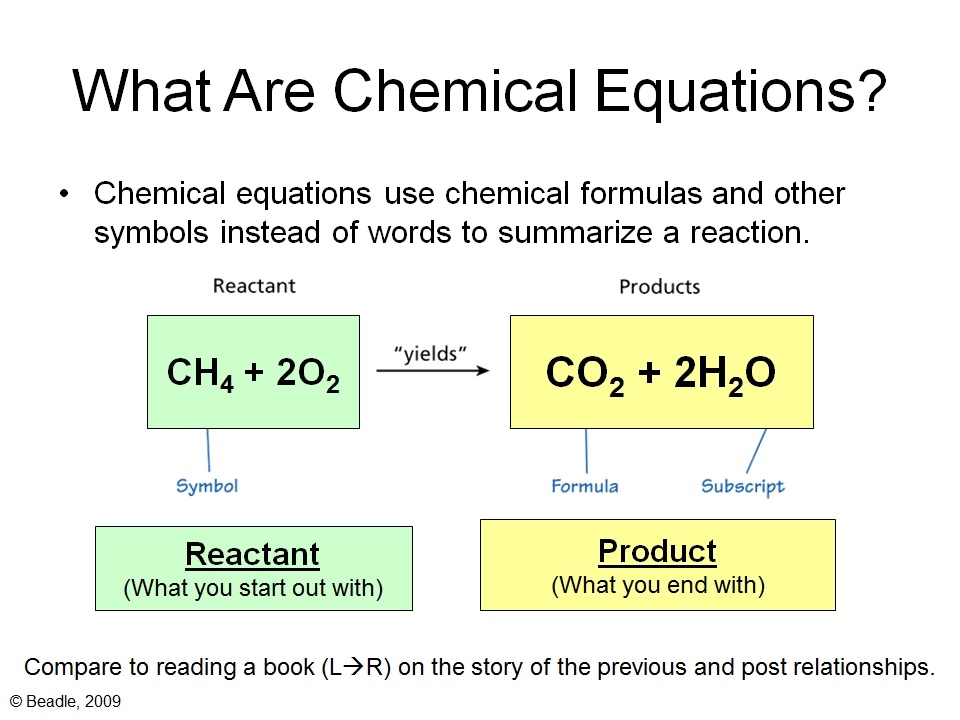
Step 2:
Balance the chemical equation by bearing in mind that the no. of atoms in reactants = no. of atoms in products. |
Question: What is the significance of balancing chemical equations?
Credit to Phet [here]